
Solutions Built to Accelerate MedTech Success
Every MedTech company’s journey is unique—but the pressure to innovate quickly, navigate regulatory hurdles, and maintain quality never changes.
Whether you’re developing a traditional medical device, IVD product, or supporting MedTech companies as a service provider, QuickVault gives you the structure and efficiency to move faster, stay compliant, and achieve commercial success.
Medical Devices
Getting a medical device to market is a race against time—but quality and compliance missteps can slow you down or stop you altogether. QuickVault unifies design, regulatory, and production workflows into a single platform, giving startups and growing teams the tools to accelerate development, secure approvals, and scale operations with confidence.
With QuickVault, medical device teams can:
- Launch faster by reducing manual processes and ensuring design controls and risk management are built-in from day one.
- Simplify regulatory submissions with submission tracking, version control, and real-time collaboration.
- Maintain quality as you scale with automated document control, supplier oversight, and training management—all validated and audit-ready.
From first concept to full-scale production, QuickVault provides the compliance foundation you need to turn innovation into commercial success.
Try it Now
In Vitro Diagnostics (IVD)
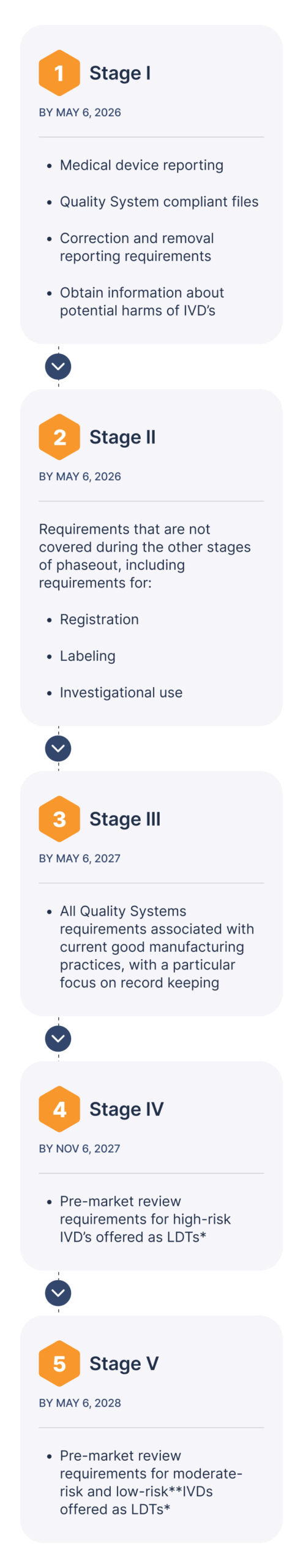
Developing and launching In Vitro Diagnostic (IVD) products comes with increasing regulatory demands. As FDA and global regulations evolve, compliance, traceability, and submission preparation more critical than ever.
QuickVault gives IVD and companies the clarity and control needed to manage compliance, simplify submissions, and accelerate time to market—without sacrificing quality.
With QuickVault, teams can:
- Accelerate product development with automated design controls and structured review processes.
- Simplify compliance documentation to meet FDA and global IVD requirements, including the latest LDT regulations.
- Maintain audit readiness through centralized records, training oversight, and integrated quality management.
From test development to commercial launch, QuickVault ensures every phase is connected, compliant, and built for speed.
Try it Now
Service Providers
Whether you’re focused on quality, regulatory, product development, or contract manufacturing, as a service provider, you are a critical partner to MedTech companies. But disconnected systems, email-based collaboration, and document versioning issues can slow projects and create risk.
QuickVault Hub transforms how you work with clients, providing a secure, shared platform to co-manage quality and regulatory processes in real time.
With QuickVault Hub, service providers can:
- Simplify collaboration with MedTech clients through dedicated QuickVault environments for each partnership.
- Ensure compliance on every project with built-in regulatory and quality workflows.
- Scale operations efficiently by streamlining document control, submission processes, and client communications.
Whether you’re advising startups or supporting commercial production, QuickVault Hub helps deliver results faster—while strengthening your competitive edge.
Learn More



Frequently Asked Questions
-
How does QuickVault support medical device companies?
QuickVault unifies design, regulatory, and production processes into a single platform, helping medical device teams streamline compliance, accelerate market entry, and scale operations efficiently. Every step is supported by built-in FDA, ISO, and GxP best practices, so you can move quickly without risking quality.
-
How does QuickVault help IVD companies manage ongoing compliance?
IVD companies face constant regulatory updates and growing expectations for traceability. QuickVault simplifies compliance by centralizing documentation, automating version control, and providing structured workflows for design, quality, and submissions. This helps IVD teams stay audit-ready while keeping development timelines on track.
-
What is QuickVault Hub for service providers?
QuickVault Hub is a shared collaboration environment that allows service providers to work directly with their MedTech clients on design, compliance, and regulatory projects—all within a validated, secure platform. This reduces inefficiencies, versioning issues, and compliance risks, enabling providers to deliver results faster and build stronger client relationships.
-
Is QuickVault suitable for startups with limited compliance experience?
Yes. QuickVault is purpose-built for MedTech startups and growing teams. The platform is pre-validated and ready to use from Day 1, with pre-built templates, guided workflows, and expert support to help teams achieve compliance—even without prior regulatory expertise.
-
Will QuickVault scale as my business grows?
Absolutely. QuickVault is designed to grow with you. Whether you’re launching your first device or expanding your product line globally, the platform adapts to support increasing complexity—without slowing you down.
The platform scales alongside your team, and our flexible subscription model ensures you always have the right level of support—without being locked into long-term contracts.
-
How does QuickVault reduce submission and approval delays?
QuickVault removes the guesswork from regulatory submissions by automating document management, ensuring version control, and providing submission tracking tools—so you can prepare faster, respond to inquiries quickly, and reduce the risk of costly delays.
-
Can QuickVault support different regulatory requirements across markets?
Yes. QuickVault is built to meet FDA, ISO, and GxP standards while also offering configurable workflows to support regional variations. Whether you’re preparing a 510(k), CE Mark submission, or aligning with MDR/IVDR, QuickVault ensures your quality and regulatory processes stay compliant.
-
How do I get started with QuickVault?
Getting started is simple. QuickVault is Day-1 ready, meaning there’s no lengthy implementation process, customization project, or hidden setup fees. You can be up and running in days—not months—saving valuable time and resources when it matters most.
Plus, our flexible, cost-effective subscription model has no long-term commitments, so you get the platform you need now, with the freedom to adjust as your company grows.
Built for Every Stage of Your MedTech Journey
No matter your focus—medical devices, IVD products, or supporting innovators as a service provider—QuickVault delivers the structure, speed, and compliance you need to succeed. From design to submission to production, our platform empowers and enables you to move faster and scale with confidence.